A chemical element is a pure chemical
substance consisting of a single type of atom distinguished by its atomic
number, which is the number of protons in its atomic nucleus. Elements are
divided into metals, metalloids, and nonmetals.
The lightest chemical elements, including hydrogen, helium and smaller amounts of lithium, beryllium and boron, are thought to have been produced by various cosmic processes during the Big Bang and cosmic ray spallation. Production of heavier elements, from carbon to the very heaviest elements, proceeded by stellar nucleosynthesis in certain planetary nebulae and supernovae, which blast these elements into space where they are available for later planetary formation in solar systems such as our own. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars.
Hydrogen and helium are by far the most abundant elements in the universe. However, iron is the most abundant element (by mass) making up the Earth, and oxygen is the most common element in Earth's crust. Although all known chemical matter is composed of these elements, chemical matter itself is hypothesized to constitute only about 15% of the matter in the universe. The remainder is believed to be dark matter, whose composition is largely unknown and most of which cannot be composed of chemical elements, since it lacks protons, neutrons or electrons.
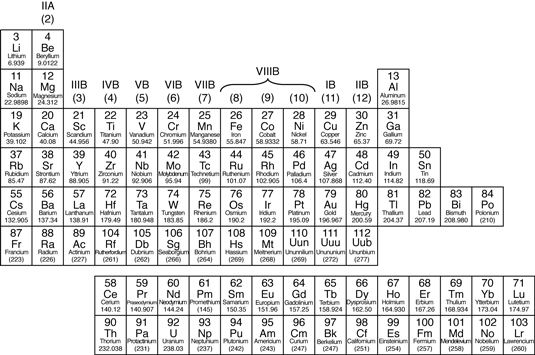
The periodic table is a tabular arrangement of the chemical elements, organized on the basis of their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. Elements are presented in order of increasing atomic number, which is typically listed with the chemical symbol in each box. The standard form of the table consists of a grid of elements laid out in 18 columns and 7 rows, with a double row of elements below that. The table can also be deconstructed into four rectangular blocks: the s-block to the left, the p-block to the right, the d-block in the middle, and the f-block below that.
The rows of the table are called periods; the columns are called groups, with some of these having names such as halogens or noble gases. Since, by definition, a periodic table incorporates recurring trends, the table can be used to derive relationships between the properties of the elements and predict the properties of new, yet to be discovered or synthesized, elements. As a result, a periodic table provides a useful framework for analyzing chemical behavior, and so the tables, in various forms, are widely used in chemistry and other sciences.
The lightest chemical elements, including hydrogen, helium and smaller amounts of lithium, beryllium and boron, are thought to have been produced by various cosmic processes during the Big Bang and cosmic ray spallation. Production of heavier elements, from carbon to the very heaviest elements, proceeded by stellar nucleosynthesis in certain planetary nebulae and supernovae, which blast these elements into space where they are available for later planetary formation in solar systems such as our own. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars.
Hydrogen and helium are by far the most abundant elements in the universe. However, iron is the most abundant element (by mass) making up the Earth, and oxygen is the most common element in Earth's crust. Although all known chemical matter is composed of these elements, chemical matter itself is hypothesized to constitute only about 15% of the matter in the universe. The remainder is believed to be dark matter, whose composition is largely unknown and most of which cannot be composed of chemical elements, since it lacks protons, neutrons or electrons.
The periodic table is a tabular arrangement of the chemical elements, organized on the basis of their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. Elements are presented in order of increasing atomic number, which is typically listed with the chemical symbol in each box. The standard form of the table consists of a grid of elements laid out in 18 columns and 7 rows, with a double row of elements below that. The table can also be deconstructed into four rectangular blocks: the s-block to the left, the p-block to the right, the d-block in the middle, and the f-block below that.
The rows of the table are called periods; the columns are called groups, with some of these having names such as halogens or noble gases. Since, by definition, a periodic table incorporates recurring trends, the table can be used to derive relationships between the properties of the elements and predict the properties of new, yet to be discovered or synthesized, elements. As a result, a periodic table provides a useful framework for analyzing chemical behavior, and so the tables, in various forms, are widely used in chemistry and other sciences.
The Periodic Law
in
chemistry, law stating that many of the physical and chemical properties of the
elements tend to recur in a systematic manner with increasing atomic number.
Progressing from the lightest to the heaviest atoms, certain properties of the
elements approximate those of precursors at regular intervals of 2, 8, 18, and
32. For example, the 2d element (helium) is similar in its chemical behavior to
the 10th (neon), as well as to the 18th (argon), the 36th (krypton), the 54th
(xenon), and the 86th (radon). The chemical family called the halogens,
composed of elements 9 (fluorine), 17 (chlorine), 35 (bromine), 53 (iodine),
and 85 (astatine), is an extremely reactive family.
"Similar properties recur periodically when elements are arranged according to increasing atomic number."
"Similar properties recur periodically when elements are arranged according to increasing atomic number."
Groups and Periods of the Periodic Table
Group (periodic table) – In chemistry, a group (also known as a family) is a
column of elements in the periodic table of the chemical elements. There are 18
numbered groups in the periodic table, but the f-block columns (between groups
2 and 3) are not numbered. The elements in a group have similar physical or
chemical characteristic of the outermost electron shells of their atoms (i.e.,
the same core charge), as most chemical properties are dominated by the orbital
location of the outermost electron. There are three systems of group numbering.
The modern numbering group 1 to group 18 is recommended by the International
Union of Pure and Applied Chemistry (IUPAC). It replaces two older naming
schemes that were mutually confusing. Also, groups may be identified by their
topmost element or have a specific name. For example, group 16 is variously
described as oxygen group and chalcogen.
Period (periodic table) – In the periodic
table of the elements, elements are arranged in a series of rows (or periods)
so that those with similar properties appear in a column. Elements of the same
period have the same number of electron shells; with each group across a
period, the elements have one more proton and electron and become less
metallic. This arrangement reflects the periodic recurrence of similar properties
as the atomic number increases. For example, the alkaline metals lie in one
group (group 1) and share similar properties, such as high reactivity and the
tendency to lose one electron to arrive at a noble-gas electronic
configuration. The periodic table of elements has a total of 118 elements.
The
Madelung energy ordering rule describes the order in which orbitals are
arranged by increasing energy according to the Madelung rule. Each diagonal
corresponds to a different value of n + l.
Modern quantum mechanics explains these periodic trends in properties in terms of electron shells. As atomic number increases, shells fill with electrons in approximately the order shown at right. The filling of each shell corresponds to a row in the table.
In the s-block and p-block of the periodic table, elements within the same period generally do not exhibit trends and similarities in properties (vertical trends down groups are more significant). However in the d-block, trends across periods become significant, and in the f-block elements show a high degree of similarity across periods.
Modern quantum mechanics explains these periodic trends in properties in terms of electron shells. As atomic number increases, shells fill with electrons in approximately the order shown at right. The filling of each shell corresponds to a row in the table.
In the s-block and p-block of the periodic table, elements within the same period generally do not exhibit trends and similarities in properties (vertical trends down groups are more significant). However in the d-block, trends across periods become significant, and in the f-block elements show a high degree of similarity across periods.
Natural Occurrence of Elements
Out
of the first 92 elements, 1 being hydrogen (H) and 92 being uranium (U), there
are 90 that are naturally occuring. Technetium (Tc) and promethium (Pm) are
man-made elements and do not have any isotopes occuring naturally. But even
though these elements occur naturally, very few are found in the elemental
state. Even fewer are in a form or in enough quantity so that one could pick
them up. Most of the elements occur only in compounds with other elements. One
of the most common compounds of elements are the oxides, which is the compound
that is produced when an element "rusts" in the presence of oxygen.
Some are very reactive with air or water and do not last long in their
elemental state, even if produced. Other elements, due to the arrangement of
the outer shell of electrons, are very reactive with other elements and are
never found out of a compound. Out of these 90 elements, 9 are gases and are usually
found in their elemental states. Since many of them are inert gases, they are
hard to find and even harder to pick up. There are 4 liquids: bromine (Br),
cesium (Cs), gallium (Ga), and mercury (Hg). Mercury is the only one found in
the elemental form in nature and only rarely and in very small amounts. There
are about 12 trace elements that do occur in their elemental form in nature,
but these are in trace amounts and you would not find them in amounts that are
recognizable. One of the elements was said to have only several grams of
elemental metal found in the crust of the earth. So that leaves the elements
that are present in big enough pieces to where you can see and pick up. These
elements are bismuth (Bi), carbon (C), copper (Cu), gold (Au), iron (Fe), lead
(Pb), manganese (Mn), silver (Ag), and sulfur (S). Each of these can be found
in veins or deposits in collectable amounts.
The answer most commonly given is 91. With the exception of the element technetium, all the elements under element 92, uranium, have been found in nature. Closer inspection shows this is not true. The number of naturally occurring elements is actually 98. Technetium is an element with no stable isotopes. It is produced artificially by bombarding samples of molybdenum with neutrons for commercial and scientific uses and is widely believed to be non-existent in nature. This has turned out to be not true. Technetium-99 can be produced when uranium-235 or uranium-238 undergoes fission. Minute amounts of technetium-99 has been found in uranium-rich pitchblende. Elements 93-98 (neptunium, plutonium, americium, curium, berkelium, and californium) were all first artificially synthesized and isolated in the Berkeley Radiation Laboratory of the University of California. They have all been found in the fallout of nuclear testing experiments and byproducts of the nuclear industry and believed to exist only in man-made forms. This also turned out to be not true. All six of these elements have been found in very small amounts in samples of uranium-rich pitchblende. This brings the total number of naturally occurring elements to 98. Perhaps one day, samples of element numbers greater than 98 will be identified.
The trite phrase "the 92 naturally-occurring chemical elements" is often seen, but is incorrect. There are only 88 naturally-occurring chemical elements. The elements 43, 61, 85 and 87 have no stable isotopes, and none of long half-life, so they are not naturally present. Small amounts are made in nuclear reactions induced by cosmic rays and nuclear tests, but these soon disappear. If you protest that these should be included, then so should Np and Pu, which are produced by the absorption of neutrons arising from spontaneous fission of uranium and thorium, and then there would be 94 naturally-occurring elements. If you wait long enough, there will only be 81 naturally-occurring elements, since everything beyond lead has only unstable isotopes, though some are of very long half-life, and have survived since the beginning, fathering their radioactive series. Any way you look at it, there are not just 92 naturally-occurring chemical elements.
The answer most commonly given is 91. With the exception of the element technetium, all the elements under element 92, uranium, have been found in nature. Closer inspection shows this is not true. The number of naturally occurring elements is actually 98. Technetium is an element with no stable isotopes. It is produced artificially by bombarding samples of molybdenum with neutrons for commercial and scientific uses and is widely believed to be non-existent in nature. This has turned out to be not true. Technetium-99 can be produced when uranium-235 or uranium-238 undergoes fission. Minute amounts of technetium-99 has been found in uranium-rich pitchblende. Elements 93-98 (neptunium, plutonium, americium, curium, berkelium, and californium) were all first artificially synthesized and isolated in the Berkeley Radiation Laboratory of the University of California. They have all been found in the fallout of nuclear testing experiments and byproducts of the nuclear industry and believed to exist only in man-made forms. This also turned out to be not true. All six of these elements have been found in very small amounts in samples of uranium-rich pitchblende. This brings the total number of naturally occurring elements to 98. Perhaps one day, samples of element numbers greater than 98 will be identified.
The trite phrase "the 92 naturally-occurring chemical elements" is often seen, but is incorrect. There are only 88 naturally-occurring chemical elements. The elements 43, 61, 85 and 87 have no stable isotopes, and none of long half-life, so they are not naturally present. Small amounts are made in nuclear reactions induced by cosmic rays and nuclear tests, but these soon disappear. If you protest that these should be included, then so should Np and Pu, which are produced by the absorption of neutrons arising from spontaneous fission of uranium and thorium, and then there would be 94 naturally-occurring elements. If you wait long enough, there will only be 81 naturally-occurring elements, since everything beyond lead has only unstable isotopes, though some are of very long half-life, and have survived since the beginning, fathering their radioactive series. Any way you look at it, there are not just 92 naturally-occurring chemical elements.
Metals, Nonmetals and Metalloids
Using the periodic table, you can
classify the elements in many ways. One useful way is by metals, nonmetals, and
metalloids. Most of the elements on the periodic table are classified as
metals.
Metals
In the periodic table, you can see a
stair-stepped line starting at Boron (B), atomic number 5, and going all the
way down to Polonium (Po), atomic number 84. Except for Germanium (Ge) and
Antimony (Sb), all the elements to the left of that line can be classified as metals.
These metals have properties that you normally associate with the metals you encounter in everyday life:
These metals have properties that you normally associate with the metals you encounter in everyday life:
- They are solid (with the exception of mercury, Hg, a liquid).
- They are shiny, good conductors of electricity and heat.
- They are ductile (they can be drawn into thin wires).
- They are malleable (they can be easily hammered into very thin sheets).
The metals in the periodic table.
Nonmetals
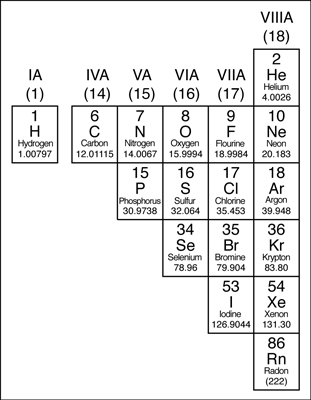
Except for the elements that border the stair-stepped line, the elements to the right of the line are classified as nonmetals (along with hydrogen). Nonmetals have properties opposite those of the metals.
The nonmetals are brittle, not malleable or ductile, poor conductors of both heat and electricity, and tend to gain electrons in chemical reactions. Some nonmetals are liquids. These elements are shown in the following figure.
The nonmetals are brittle, not malleable or ductile, poor conductors of both heat and electricity, and tend to gain electrons in chemical reactions. Some nonmetals are liquids. These elements are shown in the following figure.
The nonmetals in the periodic table.
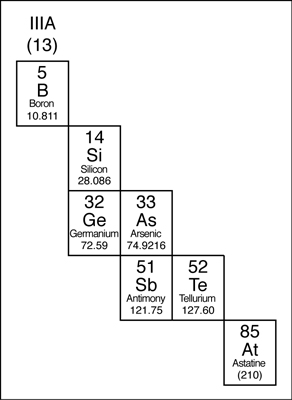
Metalloids
The elements that border the
stair-stepped line are classified as metalloids. The metalloids, or semimetals,
have properties that are somewhat of a cross between metals and nonmetals.
Metalloids tend to be economically important because of their unique conductivity properties (they only partially conduct electricity), which make them valuable in the semiconductor and computer chip industry. The metalloids are shown in the following illustration.
Metalloids tend to be economically important because of their unique conductivity properties (they only partially conduct electricity), which make them valuable in the semiconductor and computer chip industry. The metalloids are shown in the following illustration.
The metalloids in the periodic table.
Properties and Uses of Elements in the Periodic Table
Group IA: Alkali Metal –
· Sodium has many uses, it combines with chlorine to form sodium chloride, NaCl or salt, which is an essential component in preserving foods.
· Salt is mixed with crushed ice to maintain the cold temperature for “dirty ice cream” or “sorbetes”.
· Salt is also combined with 0.01% potassium iodide, KI, to manufacture iodized salt which is a common treatment for people suffering from iodine deficiency or commonly known as goiter.
· Sodium is used in the manufacture of sodium vapor lamps or commonly known as street lamps.
General properties of alkali metals:
Ø They are shiny, good conductors of heat and electricity, ductile and malleable.
Ø They are soft and can be cut by a knife.
Ø They have low density.
Ø They have relatively low melting points and boiling points compared to other metals in the Periodic Table.
Group IIA: Alkaline Earth Metals –
· Is usually combined with other metals to produce alloys or a combination of two or more metals.
· Alloys such as magnesium alloy are used in the manufacture of parts of airplanes and cars.
· Magnesium plays a vital role in the formation of chlorophyll, the green pigment in plants that captures energy from the sun to facilitate photosynthesis.
· Compounds of barium, strontium and calcium are used extensively in fireworks because when heated, it turns the flame into different colors such as pale green, crimson red and brick red.
· Oxide of calcium is used in tiles and as a lining in high tamperature furnace because of its capability to withstand high temperature.
· Calcium oxide, CaO, is used to counteract highly acidic soils so that it can be used for gardening.
· Calcium in its cationic form, Ca2+, is essential to bone health of humans and other animals.
General properties of alkaline earth metals:
Ø They are shiny, good conductors of heat and electricity, ductile and malleable.
Ø They are relatively hard.
Ø They have high density.
Ø They have relatively high melting points and boiling points.
Transition Metals –
· The elements in the coinage family are used in the productio of coins.
· Nickel, Ni, is responsible for the hardening of vegetable oil in the manufacture of margarine.
· Titanium, Ti, in the form of titanium chloride, TiCl3, is used as catalyst in the manufacture of plastics.
· Steel alloy (a combination of metals such as iron, manganese, chromium, vanadium and tungsten) are commonly used as stainless material.
· Copper ion, Cu2+, in the blood interact with pain relievers such as aspirin to relieve pain and fever.
· Cobalt, Co, is part of vitamin B12 which cures Pernicious anemia because this vitamin has the ability to increase the number of hemoglobin molecules in the blood.
General properties of transition metals:
Ø High tensile strength, density, ductility, malleability, electrical and thermal conductivity, and melting points and boiling points.
Group IVA: Carbon Family –
· Carbon exists in two allotropes, namely, diamond and graphite.
· Diamond is the hardest material known on earth which is used as abrasive, and in drilling and cuttingout other hard materials.
· The most common use of diamond is as jewelry.
· Graphite can conduct electricity that is why it isused as electrodes in batteries or dry cells.
· Silicon, the second most abundant element on earth, and germanium are widely used as semiconductors in electronic devices such as computer chips.
· Silicon is now being utilized in cosmetic medicine as body part enhancer.
· Tin are used in the manufacture of tin cans which are used in the packaging of canned foods.
· Lead is commonly used as the marking material in pencils.
Group VIIA: Halogens –
· Fluorine, in the form of fluoride, F-, is utilized extensively in the toothpaste industry. It is used to prevent tooth decay and gingivitis.
· Fluorine is utilized to make polytetrafluoroethylene, commonly known as Teflon, which is used as a cooking utensil.
· Chlorine is commonly used as a disinfectant for swimming pools.
· Chlorine is also used in industry as a bleaching agent for paper and textiles.
· Chlorine is also an ingredient in pesticides and insecticides.
· Chlorine is also a component of polyvinyl chloride or PVC which is used for pipes and ducts.
· Iodine in alcoholic solution or commonly known as tincture of iodine is widely used asantiseptic for wounds.
· Iodine is also being utilized in the production of photographic films such as silver iodide, AgI.
· Halides such as bromide, iodide, fluoride and chloride are also used in cloud seeding to catalyze the condensation of water vapor in the atmosphere.
· Sodium has many uses, it combines with chlorine to form sodium chloride, NaCl or salt, which is an essential component in preserving foods.
· Salt is mixed with crushed ice to maintain the cold temperature for “dirty ice cream” or “sorbetes”.
· Salt is also combined with 0.01% potassium iodide, KI, to manufacture iodized salt which is a common treatment for people suffering from iodine deficiency or commonly known as goiter.
· Sodium is used in the manufacture of sodium vapor lamps or commonly known as street lamps.
General properties of alkali metals:
Ø They are shiny, good conductors of heat and electricity, ductile and malleable.
Ø They are soft and can be cut by a knife.
Ø They have low density.
Ø They have relatively low melting points and boiling points compared to other metals in the Periodic Table.
Group IIA: Alkaline Earth Metals –
· Is usually combined with other metals to produce alloys or a combination of two or more metals.
· Alloys such as magnesium alloy are used in the manufacture of parts of airplanes and cars.
· Magnesium plays a vital role in the formation of chlorophyll, the green pigment in plants that captures energy from the sun to facilitate photosynthesis.
· Compounds of barium, strontium and calcium are used extensively in fireworks because when heated, it turns the flame into different colors such as pale green, crimson red and brick red.
· Oxide of calcium is used in tiles and as a lining in high tamperature furnace because of its capability to withstand high temperature.
· Calcium oxide, CaO, is used to counteract highly acidic soils so that it can be used for gardening.
· Calcium in its cationic form, Ca2+, is essential to bone health of humans and other animals.
General properties of alkaline earth metals:
Ø They are shiny, good conductors of heat and electricity, ductile and malleable.
Ø They are relatively hard.
Ø They have high density.
Ø They have relatively high melting points and boiling points.
Transition Metals –
· The elements in the coinage family are used in the productio of coins.
· Nickel, Ni, is responsible for the hardening of vegetable oil in the manufacture of margarine.
· Titanium, Ti, in the form of titanium chloride, TiCl3, is used as catalyst in the manufacture of plastics.
· Steel alloy (a combination of metals such as iron, manganese, chromium, vanadium and tungsten) are commonly used as stainless material.
· Copper ion, Cu2+, in the blood interact with pain relievers such as aspirin to relieve pain and fever.
· Cobalt, Co, is part of vitamin B12 which cures Pernicious anemia because this vitamin has the ability to increase the number of hemoglobin molecules in the blood.
General properties of transition metals:
Ø High tensile strength, density, ductility, malleability, electrical and thermal conductivity, and melting points and boiling points.
Group IVA: Carbon Family –
· Carbon exists in two allotropes, namely, diamond and graphite.
· Diamond is the hardest material known on earth which is used as abrasive, and in drilling and cuttingout other hard materials.
· The most common use of diamond is as jewelry.
· Graphite can conduct electricity that is why it isused as electrodes in batteries or dry cells.
· Silicon, the second most abundant element on earth, and germanium are widely used as semiconductors in electronic devices such as computer chips.
· Silicon is now being utilized in cosmetic medicine as body part enhancer.
· Tin are used in the manufacture of tin cans which are used in the packaging of canned foods.
· Lead is commonly used as the marking material in pencils.
Group VIIA: Halogens –
· Fluorine, in the form of fluoride, F-, is utilized extensively in the toothpaste industry. It is used to prevent tooth decay and gingivitis.
· Fluorine is utilized to make polytetrafluoroethylene, commonly known as Teflon, which is used as a cooking utensil.
· Chlorine is commonly used as a disinfectant for swimming pools.
· Chlorine is also used in industry as a bleaching agent for paper and textiles.
· Chlorine is also an ingredient in pesticides and insecticides.
· Chlorine is also a component of polyvinyl chloride or PVC which is used for pipes and ducts.
· Iodine in alcoholic solution or commonly known as tincture of iodine is widely used asantiseptic for wounds.
· Iodine is also being utilized in the production of photographic films such as silver iodide, AgI.
· Halides such as bromide, iodide, fluoride and chloride are also used in cloud seeding to catalyze the condensation of water vapor in the atmosphere.
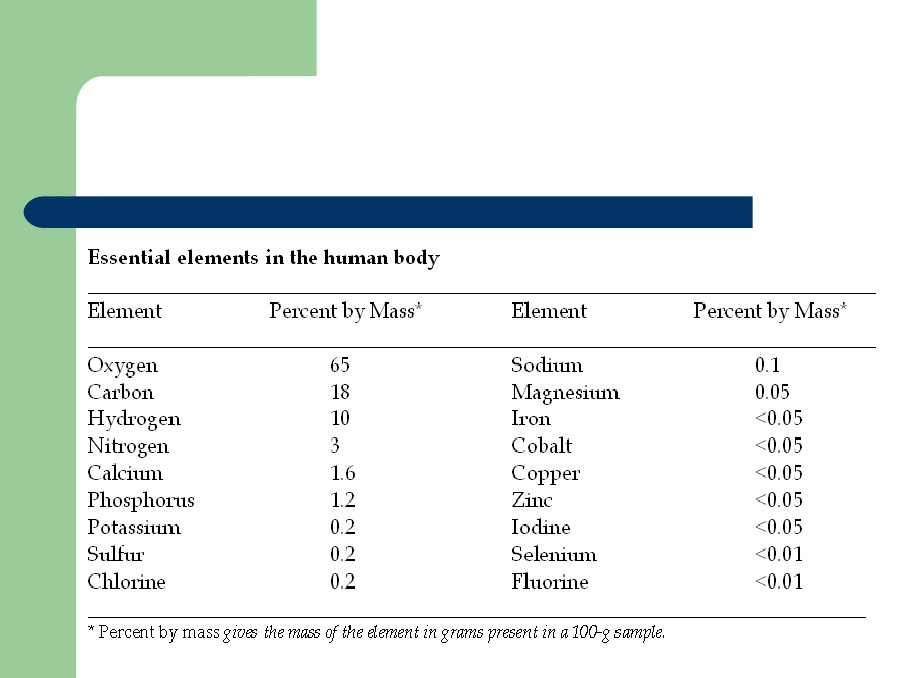
Importance of Some Elements in the Human Body
Some Elements that are Important to the Human Body –
· Trace elements such as cobalt, copper, iodine, iron and zinc actually comprises about 0.1 percent of the mass of a human body.
· The trace elements are actually necessary for biological functions like defense against sickness, transport of oxygen for metabolism and growth.
· Cobalt, copper, iron, manganese and nickel are also present in the human body. These metals aid in the catalytic activity of a variety of enzymes in the human body.
· Cytochrome oxidase, which is responsible for the burning of foods, contains copper.
· Insulin, which is responsible for the regulation of blood sugar, contains the metal chromium.
· Iron is essential to hemoglobin which carries the oxygen and dissolved foods to all parts of our body.
· The air or oxygen (O2) we breath during respiration, the water or H2O (composed of hydrogen and oxygen) we drink and some elements such as carbon, phosphorus, sodium, etc. needed to almost all physiological processes of the body are essential elements in the human body.
· Trace elements such as cobalt, copper, iodine, iron and zinc actually comprises about 0.1 percent of the mass of a human body.
· The trace elements are actually necessary for biological functions like defense against sickness, transport of oxygen for metabolism and growth.
· Cobalt, copper, iron, manganese and nickel are also present in the human body. These metals aid in the catalytic activity of a variety of enzymes in the human body.
· Cytochrome oxidase, which is responsible for the burning of foods, contains copper.
· Insulin, which is responsible for the regulation of blood sugar, contains the metal chromium.
· Iron is essential to hemoglobin which carries the oxygen and dissolved foods to all parts of our body.
· The air or oxygen (O2) we breath during respiration, the water or H2O (composed of hydrogen and oxygen) we drink and some elements such as carbon, phosphorus, sodium, etc. needed to almost all physiological processes of the body are essential elements in the human body.